Case Presentation
Case Presentation – July 2025
Medullary Thyroid Carcinoma
Written by: Joelle Tarquinio, student, Cleveland Clinic Cytology Program, Cleveland, Ohio
Patient Age: 25-year-old female
Specimen Type: Left Thyroid Bed: Smear, Pap stain, ThinPrep® Non-Gyn slide
Patient History: Patient has multiple endocrine neoplasia type 2A syndrome (MEN2A), a family history of MEN2A, and a family history of medullary thyroid carcinoma. In 2016, the patient was diagnosed with medullary thyroid carcinoma and underwent a total thyroidectomy. In 2018, the patient underwent a neck dissection due to metastasis to several lymph nodes. In September of 2024, the patient presented with a 1.3 cm mass in the left thyroid bed.
Cytologic Diagnosis: Positive for malignant cells: Medullary Thyroid Carcinoma. This diagnosis was made with comparison to the patient’s 2018 lymph node metastasis and in conjunction with the patient’s elevated calcitonin level.
Biopsy/Pathologic Diagnosis: The FNA of the left thyroid bed showed an elevated calcitonin level of 495.0 pg/ml. The tumors cells from the patient’s previous cancer showed positivity for the immunohistochemical stains CAM5.2, synaptophysin, CEA, and calcitonin.
Case provided by: Cleveland Clinic, Cleveland, Ohio
Medullary Thyroid Carcinoma
Etiology:
Unlike other thyroid tumors that stem from follicular cells, medullary thyroid carcinoma (MTC) is a neuroendocrine carcinoma that stems from the parafollicular calcitonin-producing cells of the thyroid. About 75% of MTCs arise sporadically in the 6th or 7th decade of life and are caused by RET mutations and mutations in the RAS pathway1. The remaining 25% of MTCs are hereditary and are associated with multiple endocrine neoplasia type 2 (MEN2), including the phenotypes MEN2A, MEN2B, and familial medullary thyroid carcinoma1. People with MEN2 are predisposed to develop MTC at a young age due to a germline mutation in the ret proto-oncogene2. The ret oncogene encodes the protein RET tyrosine kinase, which regulates cell proliferation, differentiation, and migration3. This is activated by cell signaling and ligand-binding. Mutations in the extracellular domain of RET tyrosine kinase can lead to amino acid substitutions that cause RET tyrosine kinase to be activated independent of ligand binding, leading to uncontrolled cell proliferation3.
Clinical Features:
MTC is relatively rare, accounting for 10% of all thyroid cancers4. Patients commonly present with an enlarged thyroid that may be accompanied by dysphagia, shortness of breath, hoarseness, and possibly lymphadenopathy1. Sporadic MTCs may be asymptomatic, causing them to present at an advanced stage4. Sporadic MTCs typically present with unilateral masses, while hereditary MTC masses are typically bilateral5. Patients who have MEN2 or a family history of MTC /MEN2 have a high risk of developing hereditary MTC and may undergo routine MTC screening1. MTC is often accompanied by elevated levels of calcitonin in blood, reflecting the carcinoma’s calcitonin-producing activity6. Rarely, MTC may secrete adrenocorticotropin releasing hormone or corticotropin releasing hormone, leading to the development of Cushing’s disease6.
Treatment and Prognosis:
Treatment of MTC is often individualized, dependent on stage, type, and specific genetic mutation. Genetic testing is recommended for all cases of MTC to identify mutations that may have targeted therapies and to guide surgical management1. People with high-risk germline RET mutations are candidates for prophylactic thyroidectomy to prevent the development of MTC1. When the tumor is confined to the thyroid, the most standard treatment is surgical resection. MTCs diagnosed following lobectomy are not followed by total thyroidectomy unless the MTC is determined to be hereditary4. Due to the multifocal nature of hereditary MTCs, complete thyroidectomy is recommended to prevent recurrence. Neck dissection may be performed when there is metastasis to the lymph nodes and can be considered when the tumor measures larger than one centimenter4. Treatments for recurrent or residual MTCs include further surgical resection, radiation, ablation, chemotherapy, and kinase inhibitors4.
Patients diagnosed with MTC show variation in prognosis. The five-year and ten-year survival rates of MTC are 65-90% and 45-80%, respectively6. Factors that correlate to a poor prognosis included older age, cervical node metastasis, being male, having MEN 2B higher-grade tumor, and specific RET mutations6. Favorable prognostic factors include younger age, being female, and confinement to the thyroid6.
Cytology:
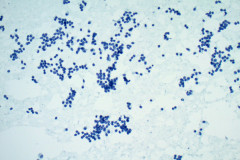
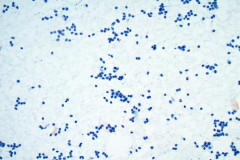
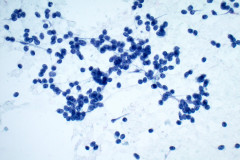
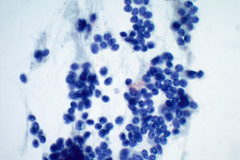
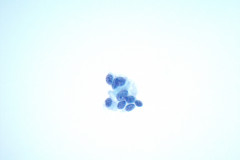
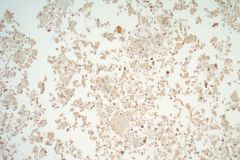
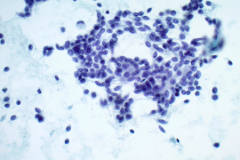
Aspirates of MTC are moderately cellular, showing a single cell pattern with occasional clustering and small aggregates. Cytologically, tumor cells can appear plasmacytoid, polygonal, or with spindle cells. Cells are uniform in size, but rare large pleomorphic cells may be present. The chromatin shows a coarse, granular, neuroendocrine pattern with inconspicuous nucleoli. Intranuclear inclusions may be seen, and nuclear grooves are rare. The cytoplasm is granular, and the amount is variable and rarely scant. Cytoplasmic dendritic processes may also be seen. Amyloid may be present in the background, although it can be identical to colloid in Papanicolaou-stained specimens. Variants of MTC include a follicular variant, papillary variant, small cell variant, giant cell variant, and a clear cell variant7. Cell blocks of MTC typically stain positive for calcitonin, CEA, TTF1, synaptophysin, and chromogranin, and are negative for thyroglobulin and PAX-88,9.
The tumor cells of this case presentation posed a diagnostic challenge due to their divergence from the typical cytological appearance of MTC. The majority of tumor cells had scant cytoplasm, including cells that resembled naked nuclei and cells with a plasmacytoid appearance. A diagnosis of MTC was rendered in conjunction with the patient’s elevated calcitonin level, history of MTC and MEN2, and comparison to the patient’s previous two MTC diagnoses.
Differential Diagnosis:
The diverse appearance and cytomorphological criteria of MTC overlap with that of other neoplasms, leading to diagnostic challenges. These challenges may stem from MTC’s variable cellular patterns, chromatin pattern, cell clustering, and presence or lack of plasmacytoid cytoplasm8. Differential diagnoses of MTC include Anaplastic Thyroid Carcinoma, Papillary Thyroid Carcinoma, and metastatic neuroendocrine carcinoma.
Misinterpretation of MTC as Anaplastic Thyroid Carcinoma (ATC) may occur with the presence of large pleomorphic cells or spindle cells, which are characteristic of ATC9. While not a characteristic appearance, plasmacytoid cells may also occur in ATC, causing even more cytologic overlap8. Similar to MTC, ATC aspirates are also moderately cellular, exhibiting isolated cell patterns with small aggregates. Cytologic features of ATC that MTC typically lacks include extreme pleomorphism, prominent nucleoli, necrosis, and increased mitotic activity8. The immunohistochemical profile of ATC includes positivity for PAX8, and negativity for TTF1, thyroglobulin, and neuroendocrine markers8. MTC stains positive for neuroendocrine markers and calcitonin can be helpful in making its diagnosis.
Papillary Thyroid Carcinoma (PTC) is a differential of MTC because of their overlapping features and the rare papillary variant of MTC7. MTC can present with intranuclear inclusions which may mimic PTC. However, the neuroendocrine chromatin and lack of nuclear grooves helps to distinguish MTC from PTC cytologically. The MTC papillary variant exhibits papillary growth associated with fibrovascular stalks, which is characteristic of PTC7. In all cases of MTC, immunohistochemical positivity for calcitonin and neuroendocrine markers should rule out PTC in diagnosing MTC malignancies.
The characteristic neuroendocrine chromatin seen in MTC may lead to a differential diagnosis of metastatic neuroendocrine carcinomas. MTC can exhibit scant cytoplasm and uniform nuclei, along with neuroendocrine chromatin, mimicking small cell carcinoma8. Also, the plasmacytoid nature of MTC may resemble plasmacytoid carcinoid cells. Small cell carcinoma and carcinoid tumors both have immunohistochemical positivity for neuroendocrine markers, leaving positivity for calcitonin as a strong support of a MTC diagnosis.
References:
- Ramos Santillan V, Master SR, Menon G, et al. Medullary Thyroid Cancer. [Updated 2024 Nov 10]. In:StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459354/
- Eng C, Plitt G. Multiple Endocrine Neoplasia Type 2. 1999 Sep 27 [Updated 2023 Aug 10]. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1257/
- A Taccaliti, F Silvetti, G Palmonella, M Boscaro. Genetic alterations in medullary thyroid cancer: diagnostic and prognostic markers. Curr Genomics. 2011 Dec;12(8):618- 25. doi: 10.2174/138920211798120835. PMID: 22654561; PMCID:PMC3271314.
- Kim M, Kim BH. Current Guidelines for Management of Medullary Thyroid Carcinoma. Endocrinol Metab (Seoul). 2021 Jun;36(3):514-524. doi: 10.3803/EnM.2021.1082. Epub 2021 Jun 22. PMID: 34154310; PMCID: PMC8258323.
- Matias-Guiu, X., De Lellis, R. Medullary Thyroid Carcinoma: a 25-Year Perspective. Endocr Pathol 25, 21–29 (2014). https://doi.org/10.1007/s12022-013-9287-2
- Wei S. Medullary thyroid carcinoma. PathologyOutlines.com website. https://www.pathologyoutlines.com/topic/thyroidmedullary.html. Accessed February 7th, 2025.
- Carcangiu, M., Delellis, R., Rosai, J. Atlas of Tumor Pathology: Tumors of the Thyroid Gland, Third Series, Fascicle 5. Armed Forces Institute of Pathology, Washington, D.C. 1990.
- Ali SZ, Cibas ES. The Bethesda System for Reporting Thyroid Cytopathology: Definitions, Criteria, and Explanatory Notes. Cham: Springer; 2018.
- Cibas ES, Ducatman BS. Cytology: Diagnostic Principles and Clinical Correlates. 5th Ed. Philadelphia, PA: Elsevier; 2021.